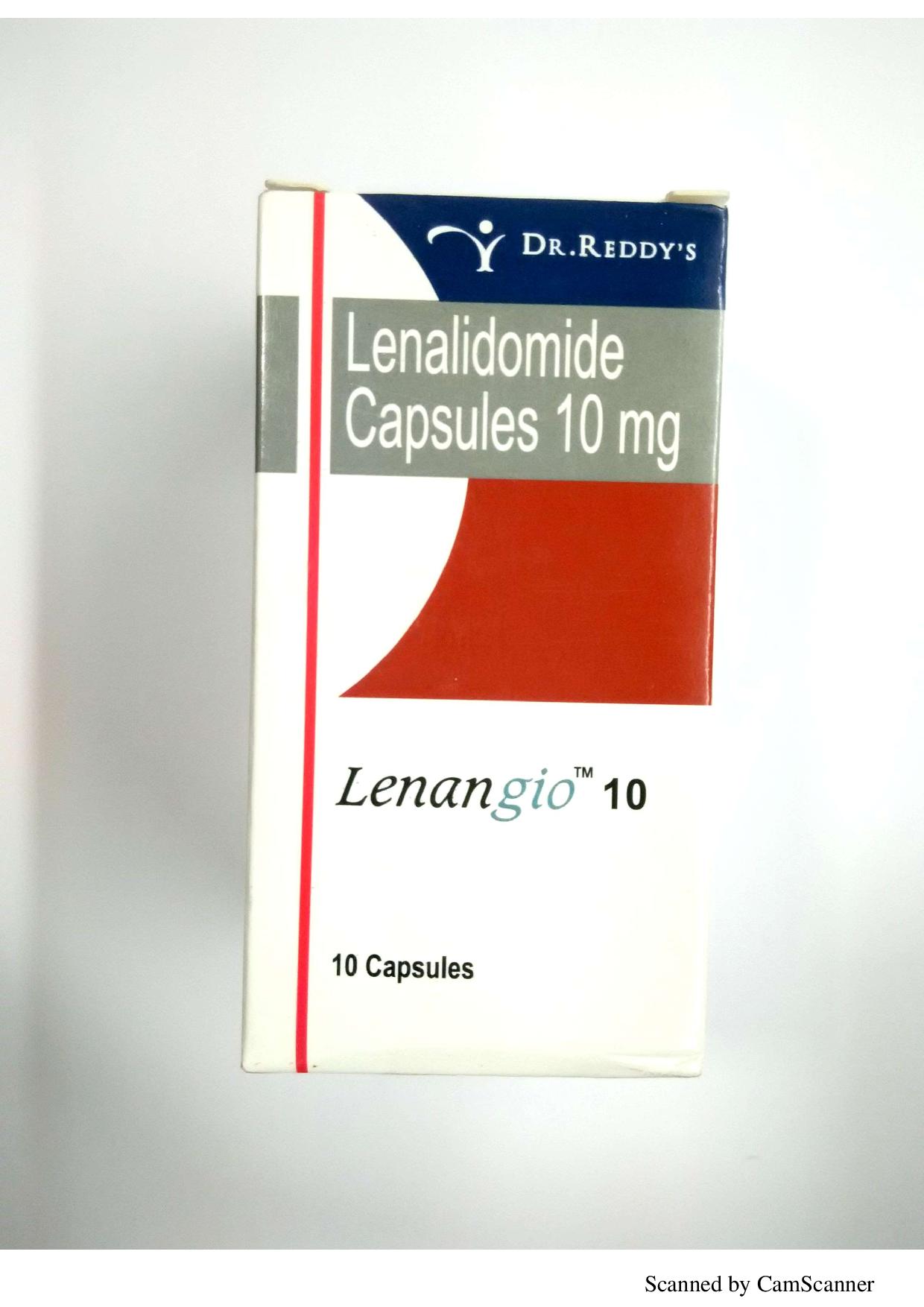
DESCRIPTION
LENANGIO (Lenalidomide ) is used in the treatment of multiple myeloma and lepra reaction. It modulates the activity of body’s immune system and indirectly helps in attacking the cancer cells. This drug is used to treat people with a disease of the bone marrow called myelodysplastic syndrome. It is also sometimes used to treat multiple myeloma and is being studied for use against other cancers. Lenangio is a prescription drug.
Manufactured : Dr Reddy’s Laboratories Ltd
ADDITIONAL INFORMATION
Strengths available : 5mg , 10mg, 25mg Capsules
Storage : Store in a cool place, protected from moisture
Dosage :
Lenangio should be taken orally at about the same time each day, either with or without food. Lenangio capsules should be swallowed whole with water. The capsules should not be opened, broken, or chewed.
Multiple Myeloma : The recommended starting dose of Lenangio is 25 mg orally once daily on Days 1-21 of repeated 28-day cycles in combination with dexamethasone.
In patients who are not eligible for autologous stem cell transplantation (ASCT), treatment should continue until disease progression or unacceptable toxicity.
For patients who are ASCT-eligible, hematopoietic stem cell mobilization should occur within 4 cycles of a Revlimid-containing therapy.
SIDE EFFECTS
Most common side effects are Headache, Weakness, Nausea, Rash, Breathlessness, Dizziness, Sleepiness, Edema, Loss of appetite, Decreased white blood cell count, Decreased calcium level in blood, Weight gain, Muscle weakness, Fatigue, Fever, Anxiety, Blood clots , Dry skin, Weight loss, Confusion, Decreased white blood cell count (neutrophils), Neuropathy, Constipation, Tremor.
PACK SIZE
10 capsules per bottle
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of LENANGIO Lenalidomide 5mg , 10mg & 25mg Capsules based in India - To buy LENANGIO Lenalidomide 5mg , 10mg & 25mg Capsules or know its cost price contact us here.
We supply & sell LENANGIO Lenalidomide 5mg , 10mg & 25mg Capsules for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.