Disclaimer
All trademarks, logos and brand names are the property of their respective owners, used here solely for identification & reference only under Section 30(1) of the Trade Marks Act, 1999. We are not affiliated with or endorsed by them unless explicitly stated. By using this website, you agree to these terms.
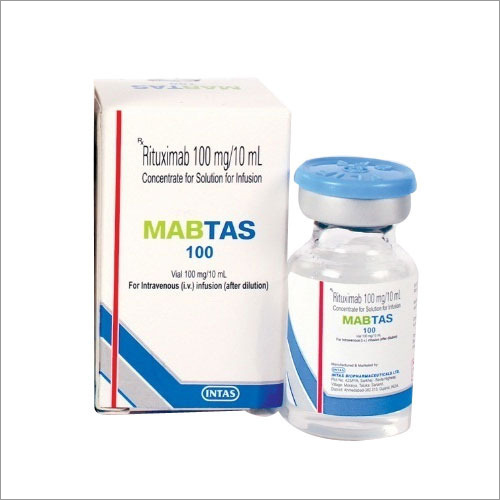
MABTAS (Rituximab) belongs to a group of cancer drugs known as monoclonal antibodies . MABTAS is used to treat several types of CD20-positive NHL (non-Hodgkin lymphoma ). It may also be used to treat blood cancer -CLL (Chronic lymphocytic leukemia) and Rheumatoid arthritis. This drug is administered only as an IV infusion. MABTAB is a prescription drug .
Manufacturer : Intas Pharmaceuticals Ltd
ADDITIONAL INFORMATION
Strengths available : 100mg & 500mg
Dosage :
- Administer only as an Intravenous Infusion.
- Do not administer as an intravenous push or bolus.
- Premedicate before each infusion.
Mabtas should only be administered by a healthcare professional with appropriate medical support to manage severe infusion reactions that can be fatal if they occur.
First Infusion: Initiate infusion at a rate of 50 mg/hr. In the absence of infusion toxicity, increase infusion rate by 50 mg/hr increments every 30 minutes, to a maximum of 400 mg/hr.
Subsequent Infusions: Initiate infusion at a rate of 100 mg/hr. In the absence of infusion toxicity, increase rate by 100 mg/hr increments at 30-minute intervals, to a maximum of 400 mg/hr.
SIDE EFFECTS
Most common side effects are Headache, Weakness, Edema, Infection, Hair loss, Itching, Chills, Febrile neutropenia, Decreased white blood cell count (neutrophils), Infusion reaction.
PACK SIZE
Pack of 1 Vial
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of MABTAS Rituximab 100mg & 500 based in India - To buy MABTAS Rituximab 100mg & 500 or know its cost price contact us here.
We supply & sell MABTAS Rituximab 100mg & 500 for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.