Disclaimer
All trademarks, logos and brand names are the property of their respective owners, used here solely for identification & reference only under Section 30(1) of the Trade Marks Act, 1999. We are not affiliated with or endorsed by them unless explicitly stated. By using this website, you agree to these terms.
Description
Additional Information
Side Effects
Pack Size
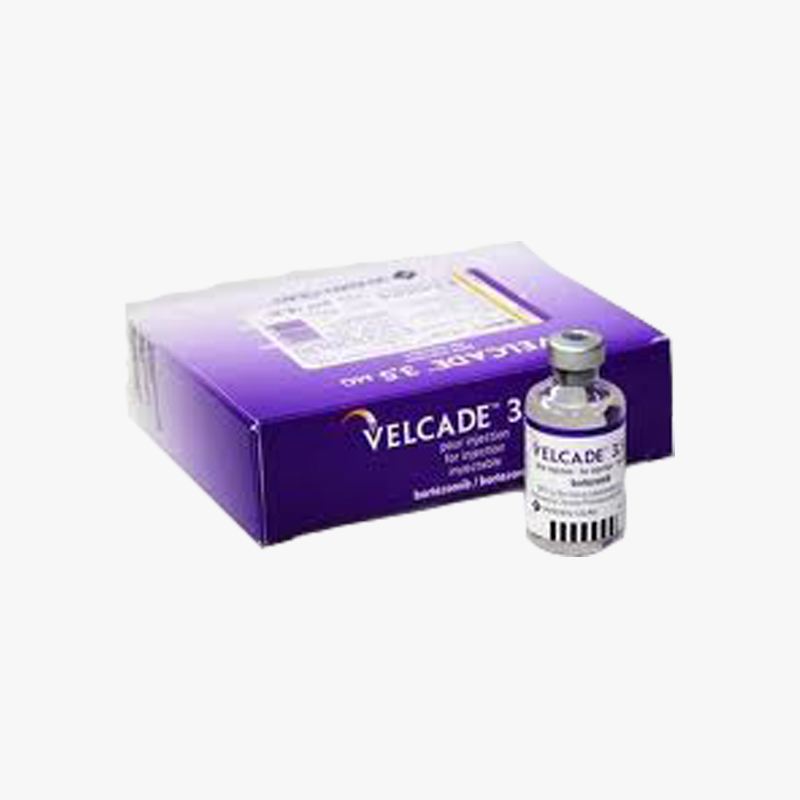
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of Velcade 3.5 mg Injection based in India - To buy Velcade 3.5 mg Injection or know its cost price contact us here.
We supply & sell Velcade 3.5 mg Injection for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.